FDA: Global Pharma's eye drops contaminated with “filth” while made unapproved claims for eye drops

PDF) Content Analysis of US FDA Warning Letters Issued to Compounding Pharmacies Regarding Violations of Current Good Manufacturing Practices Between 2017 and 2022

Complete Abstract book (15.8MB PDF) - ISID Home - International

Afrilabs – Visa Foundation: $100,000 USD for women entrepreneurs

PDF) The Dawn of Drug safety Myles Stephens

a4cd9060-5d25-11e1-bc69-0016367650a7

Arthritis Supplements Reviewed by

BEYC #673 - Bored Eye Yawn Club

Cosmeceuticals
Latest Information About Eye Health Supplements: Product Reviews, Warnings, Recalls, & Clinical Updates from
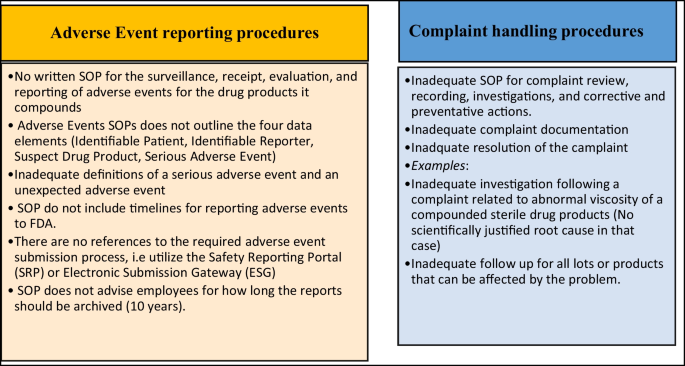
Content Analysis of US FDA Warning Letters Issued to Compounding Pharmacies Regarding Violations of Current Good Manufacturing Practices Between 2017 and 2022









